Publications of the laboratory
Group of Dr. Mikhail Beklemishev
Main research topics:
- Fluorescent fingerprinting techniques (pattern-based sensing): reaction-based fingerprinting, "fluorescent tongue", "fluorescent eye"
- Aggregation-based fluorescence methods: visualization of hydrophobic ion pairs (HIPs)
- Aggregation-based imaging of hydrophilic small molecules in biological samples using NIR fluorophores
Research Topics
Fluorescent Tongue (Fluorescent Eye)
- Since 2017 we have been developing a qualitative chemical analysis technique named “fluorescent tongue” (“eye”) based on the interaction of the components of the analyzed solution with a set of fluorophores of various nature, the emission intensity of which changes due to the compounds present in the sample. The nature of these substances is not known (it can be studied, but it is a separate task). Thus, “fluorescent tongue” refers to the so-called “fingerprinting” methods, in which any “picture” (a spectrum, chromatogram or voltammogram) is obtained from the sample, which is then processed as a whole, without identifying the components, by chemometric methods (principal component analysis (PCA), linear discriminant analysis (LDA) etc).
96-well plates with various fluorophores used in the discrimination of model drug mixtures (c, a, s, p = chloramphenicol, amikacin, sulfamethoxazole, pyracetam; o = no analyte) with fluorescein attached to the surface of silica nanoparticles via poly(ethyleneimine) (left plate) and a mixture of 4 fluorophores (right plate). Irradiation by a 254-nm lamp in Camag Visualizer 2, exposure time 10 sec.
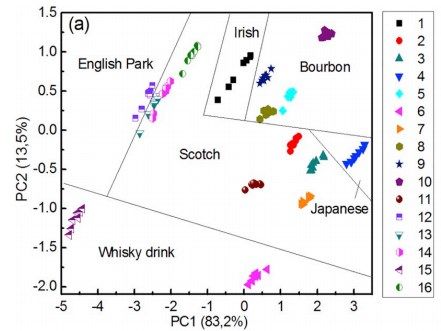
Classification of whiskey by type using one fluorophore, Ru(bpy)3)2+
Reaction-base Sensing
- In 2021 we started to develop a fluorescence platform based on slow indicator reactions for the purposes of fingerprinting. In this case, the sample is mixed with the dye, an oxidizer and a catalyst of oxidation (for instance, hydrogen peroxide and Cu(2+) salt). The catalyst and the intermediate particles can interact with the components of the sample, thereby changing the reaction rate, which makes the system sensitive to the composition of samples. Reaction rate is controlled by periodical photographing the 96-well plate with a NIR photo camera upon 660-nm LED excitation and/or 254/366-nm excited visible fluorescence. Absorbance can be also measured using photographs in visible light. Aggregation principles are also employed in the kinetic-based fluorescence platform.
Abstracts of papers
Optical sensing is an inexpensive and simple technique in water pollution control that offers high sample throughput and field applications. In this work, we applied an innovative reaction-based optical fingerprinting strategy for the recognition of clean and synthetically contaminated waters. Samples of spring, well, borehole, pond and tap water were introduced into the reaction mixtures containing carbocyanine dyes, the absorbance and fluorescence of which were monitored photographically at different times; the data were processed by linear discriminant analysis. Up to 11 clean water samples were completely discriminated; the samples artificially polluted with the soluble fraction of different brand engine oil (WSF) and iron(2+) supplied as Mohr’s salt were discriminated from uncontaminated ones; the presence of 35-fold diluted WSF in water was also detected. Iron(2+) was detected in water samples at 3 µg/L using the same indicator reactions. The proposed strategy can be useful in water pollution monitoring.
Evgenii V. Skorobogatov, Anna V. Shik, Pavel V. Sobolev, Irina A. Stepanova, Vladislav S. Orekhov, Alexander O. Ustyuzhanin, Marina V. Koksharova, Yury A. Ikhalaynen, Yury V. Timchenko, Igor A. Rodin, Mikhail K. Beklemishev. Monitoring Different Water Types for Engine Oil Water-Soluble Fraction and Iron(2+) Using a Reaction-Based Optical Sensing Strategy: a Proof-of-Concept Study. Industrial & Engineering Chemistry Research, 2024, DOI: 10.1021/acs.iecr.4c01502
Food irradiation is becoming increasingly popular in many countries for preserving and extending the shelf life of foods, which creates a demand for express methods for the detection of absorbed doses. This paper proposes an innovative method for the estimation of the dose absorbed by X-rayed beef samples using a reaction-based optical sensing technique that was proposed earlier by our team for the estimation of the dose absorbed by raw potatoes. Potato and beef samples were exposed to X-ray irradiation at 100 and 1000 Gy and then extracted by water at 23°C for 24 h or 60/70°C for 1 h. The resulting solutions were introduced to the reaction mixtures of dyes (carbocyanines, Rhodamines, or Crystal Violet) with oxidants (hypochlorite, bromate, or hydrogen peroxide). The fluorescence intensity and absorbance of the mixtures were periodically measured photographically using visualizers. The data were processed using linear discriminant analysis (LDA) and k-nearest neighbors algorithm (kNN). Using the most efficient individual reactions, the doses can be recognized with a 90–100% accuracy, and the combinations of up to 5 reactions can improve the accuracy to 100%. The protocol is simple and rapid: sample extraction time and indicator reactions take not more than 1 h each. The proposed method potentially has a wide area of applications ranging from plants to animal products.
Shik, A.V.; Sobolev, P.V., Zubritskaya, Y.V.; Baytler, M.O.; Stepanova, I.A.; Chernyaev, A.P.; Borschegovskaya, P.Yu.; Zolotov, S.A.; Doroshenko, I.A.; Podrugina, T.A.; Bliznyuk, U.A.; Rodin, I.A.; Beklemishev, M.K. Rapid Testing of Irradiation Dose in Beef and Potatoes by Reaction-Based Optical Sensing Technique, Journal of Food Composition and Analysis, 127, 2024, Article 105946. https://doi.org/10.1016/j.jfca.2023.105946 (IF = 4.52, Q1).
Array-based optical sensing is an efficient technique for the determination and discrimination of small organic molecules. This study is aimed at the development of a simple and rapid strategy for obtaining an optical response from a wide range of low-molecular-weight organic compounds. We have suggested a colorimetric and fluorimetric sensing platform based on the combination of two response mechanisms using carbocyanine dyes: aggregation and oxidation. In the first one, the analyte forms ternary aggregates with an oppositely charged surfactant wherein the dye is solubilized in the hydrophobic domains of the surfactant accompanied with fluorescent enhancement. The second mechanism is based on the effect of the analyte on the catalytic reaction rate of dye oxidation with H2O2 in the presence of a metal ion (Cu2+, Pd2+), which entails fluorescence waning and color change. The reaction mixture in a 96-well plate is photographed in visible light (colorimetry) and near IR region under red light excitation (fluorimetry). In this proof-of-concept study we demonstrated the feasibility of discrimination of 9 medicinal compounds using principle component analysis: 4 cephalosporins (ceftriaxone, cefazolin, ceftazidime, cefotaxime), 3 phenothiazines (promethazine, promazine, chlorpromazine), and 2 penicillins (benzylpenicillin, ampicillin) in an aqueous solution and in the presence of turkey meat extract. The suggested platform allows simple and rapid recognition of analytes of various nature without using spectral equipment, except for a photo camera.
Shik, A.V.; Stepanova, I.A.; Doroshenko, I.A.; Podrugina, T.A.; Beklemishev, M.K. Carbocyanine-Based Fluorescent and Colorimetric Sensor Array for the Discrimination of Medicinal Compounds. Chemosensors 2022, 10, 88. https://doi.org/10.3390/chemosensors10020088
Optical sensor arrays are widely used in obtaining fingerprints of samples, allowing for solutions of recognition and identification problems. An approach to extending the functionality of the sensor arrays is using a kinetic factor by conducting indicator reactions that proceed at measurable rates. In this study, we propose a method for the discrimination of proteins based on their oxidation by sodium hypochlorite with the formation of the products, which, in turn, feature oxidation properties. As reducing agents to visualize these products, carbocyanine dyes IR-783 and Cy5.5-COOH are added to the reaction mixture at pH 5.3, and different spectral characteristics are registered every several minutes (absorbance in the visible region and fluorescence under excitation by UV (254 and 365 nm) and red light). The intensities of the photographic images of the 96-well plate are processed by principal component analysis (PCA) and linear discriminant analysis (LDA). Six model proteins (bovine and human serum albumins, γ-globulin, lysozyme, pepsin, and proteinase K) and 10 rennet samples (mixtures of chymosin and pepsin from different manufacturers) are recognized by the proposed method. The method is rapid and simple and uses only commercially available reagents.
Shik, A.V.; Stepanova, I.A.; Doroshenko, I.A.; Podrugina, T.A.; Beklemishev, M.K. Carbocyanine-Based Optical Sensor Array for the Discrimination of Proteins and Rennet Samples Using Hypochlorite Oxidation. Sensors 2023, 23, 4299. https://doi.org/10.3390/s23094299
High-energy electron beam and X-ray processing of foods can be used for extending their storage life and for combating pests and pathogens. Several instrumental techniques are used to estimate irradiation doses in foods, but these methods are complex and laborious, require expensive equipment, and do not always allow to determine low doses. This study was aimed at developing simple methods for detecting irradiation in potato tubers and for dose estimation. We used a “fingerprinting” strategy that does not involve quantitation of any compound; instead, the rate of indicator reactions involving carbocyanine dyes is measured. The dye content was monitored by its near-infrared fluorescence intensity and visible-light absorption. Potatoes not subjected to treatment and those irradiated with different doses (10, 100, 1000, 5000, or 10,000 Gray) could be distinguished by linear discriminant analysis. Thus, the order of magnitude of the absorbed dose can be estimated with 89% ± 3% accuracy for a mixture of tubers of two potato varieties irradiated with an electron beam or with 95% ± 8% accuracy for one variety irradiated with an X-ray source.
Anna V. Shik, Evgenii V. Skorobogatov, Ulyana A. Bliznyuk, Alexander P. Chernyaev, Valentina M. Avdyukhina, Polina Yu. Borschegovskaya, Sergey A. Zolotov, Maksim O. Baytler, Irina A. Doroshenko, Tatyana A. Podrugina, Mikhail K. Beklemishev. Estimation of doses absorbed by potato tubers under electron beam or X-ray irradiation using an optical fingerprinting strategy. Food Chemistry, 414 (2023) Article number 135668, DOI: 10.1016/j.foodchem.2023.135668.
Optical “fingerprints” are widely used for chemometrics-assisted recognition of samples of different types. An emerging trend in this area is the transition from obtaining “static” spectral data to reactions analyzed over time. Indicator reactions are usually carried out in aqueous solutions; in this study, we developed reactions that proceed in an organic solvent, thereby making it possible to recognize fat-soluble samples. In this capacity, we used 5W40, 10W40, and 5W30 motor oils from four manufacturers, with six samples in total. The procedure involved mixing a dye, sample, and reagents (HNO3, HCl, or tert-butyl hydroperoxide) in an ethanolic solution in a 96-well plate and measuring absorbance or near-infrared fluorescence intensity every several minutes for 20–55 min. The obtained photographic images were processed by linear discriminant analysis (LDA) and the k-nearest neighbors algorithm (kNN). Discrimination accuracy was evaluated by a validation procedure. A reaction of oxidation of a dye by nitric acid allowed us to recognize all six samples with 100% accuracy for LDA. Merging of data from the four reactions that did not provide complete discrimination ensured an accuracy of 93% for kNN. The newly developed indicator systems have good prospects for the discrimination of other fat-soluble samples. Overall, the results confirm the viability of the kinetics-based discrimination strategy.
Pypin, A.A.; Shik, A.V.; Stepanova, I.A.; Doroshenko, I.A.; Podrugina, T.A.; Beklemishev, M.K. A
Reaction-Based Optical Fingerprinting Strategy for the Recognition of Fat-Soluble Samples: Discrimination of Motor Oils. Sensors 2023, 23, article no. 7682. https://doi.org/10.3390/s23187682
Fluorescence imaging
- The development of imaging methods involves obtaining a pattern of the distribution of a low molecular weight organic compound by the fluorescence technique. The need for imaging, rather than determining the gross content, arises while solving medicinal problems and in the study of biological samples. Imaging techniques are well developed for biomolecules and inorganic ions, but for “small molecules” such methods are well developed for sulfur compounds (2–), nitro compounds, and a few other substances. The most convenient way of imaging is luminescence using fluorophores, the excitation and emission of which are related to the red and near infrared (NIR) regions of the spectrum.
Confocal microscope images of the aggregates dye–poly(hexamethylene guanidine) (PHMG) and dye–ceftriaxone–PHMG encapsulated into different chitosan types endocytosed into cells. Images from left to right show:
channel 1 (λex = 515 nm), channel 2 (λex = 635 nm), and an overlay.
Aggregation-based methods using carbocyanine dyes
This study is aimed at developing sensing schemes without obtaining selective receptors. A series of simple carbocyanine dyes was synthesized, whose emission was quenched in water with formation ofnanoparticles in the range of 20–100 nm. Fluorescence in near-IR region is ‘‘turned on” in the presence of a drug cation of middle molecular weight (400–700 Da) and sodium dodecyl sulfate (SDS), as well as anionic drugs and a cationic surfactant (cetyltrimethylammonium bromide, CTAB). Aggregates (clusters) up to 100–200 nm in size were detected using dynamic light scattering (DLS) and Rayleigh light scattering (RLS) techniques in the systems: cationic analyte–SDS, carbocyanine dye–CTAB, and in all brightly fluorescent ternary systemsdye–surfactant–analyte. Small ions (<200 Da) incapable of multi-point binding do not form the aggregates or cause the emission enhancement. The ‘‘turn-on” signal is only observed at the surfactant submicellar concentrations insufficient to solubilize the dye nanoparticles. Based on these findings, we suggest a rapid and simple method for the detection of 4·10-5 mol/L of neomycin in urine. The proposed strategy paves the way for developing more selective methods.
Zakharenkova, S.A.; Katkova, E.A.; Doroshenko, I.A.; Kriveleva, A.S.; Lebedeva, A.N.; Vidinchuk, T.A.; Shik, A.V.; Abramchuk, S.S.; Podrugina, T.A.; Beklemishev, M.K. Aggregation-based fluorescence amplification strategy: "turn-on" sensing of aminoglycosides using near-IR carbocyanine dyes and pre-micellar surfactants. Spectr. Acta A, 247, 2021, 119109. DOI: 10.1016/j.saa.2020.119109
https://www.sciencedirect.com/science/article/abs/pii/S138614252031088X
Methods of selective fluorescent sensing of small organic molecules require the use of biomolecules or complicated synthesis of artificial probes. An alternative technique is aggregation-based sensing based on the response generated due to self-assembly effects rather than by direct analyte–probe interaction. We have found that far red/near IR emission of spinach chlorophyll a at 680 nm is selectively enhanced by multiply charged aminoglycoside cations in the presence of anionic surfactants, whereas such an effect is not observed for smaller species. Selectivity of the detection is controlled by a positive charge of the drug and its ability to form multiple noncovalent bonds, as well as by the nature of the surfactant (n-dodecyl sulfate or lauroyl sarcosinate is preferable). Fluorescence of chlorophyll is enhanced due to its solubilization in the drug–surfactant aggregate. We have developed a simple and selective method for the detection of aminoglycoside antibiotic neomycin in urine samples with a limit of detection of 6 μM. The development path for aggregation-based analysis of other systems using chlorophyll is outlined.
Sofia A. Zakharenkova, Andrey A. Dobrovolskii, Alexey V. Garshev, Mikhail A. Statkus, and Mikhail K. Beklemishev*. Chlorophyll-Based Self-Assembled Nanostructures for Fluorescent Sensing of Aminoglycoside Antibiotics. ACS Sustainable Chem. Eng. 2021, 9, 9, 3408–3415. https://doi.org/10.1021/acssuschemeng.0c08223
Imaging-guided delivery is developed for hydrophobic drugs, and to a much lesser extent, hydrophilic ones. In this work we have designed a novel strategy for real-time monitoring of hydrophilic drug delivery. Traditionally, the drug and the dye are covalently attached to a nanocarrier or are electrostatically adsorbed. Recently, we found an efficient way to bind the drug by ion-paring with an appropriate counter-ion to form the aggregate that embeds a hydrophobic dye with a considerable fluorescence enhancement. We synthesized a series of carbocyanine dyes of hydrophobicity sufficient for solubilization in hydrophobic ion pairs, which restores their emission in the near-infrared (NIR) region upon the formation of the ternary aggregates. To avoid using toxic surfactants, we applied an amphiphilic polymer-oligomer poly(hexamethylene guanidine) (PHMG) as a counter-ion. Сeftriaxone was used as a model hydrophilic drug ensuring the highest fluorescent signal. The so-formed drug–counter-ion–dye aggregates were encapsulated into a cross-linked maleated chitosan carrier. Confocal laser scanning microscopy (CLSM) studies have demonstrated internalization of the encapsulated model drug by breast adenocarcinoma cells at 40 min after treatment. These results suggest the potential application of hydrophobic ion pairs containing an NIR dye in imaging-guided delivery of hydrophilic compounds.
Zakharenkova, S.A.; Lebedeva, M.I.; Lebedeva, A.N.; Doroshenko, I.A.; Vlasova, K.Y.; Bartoshevich, A.A.; Senyavin, V.M.; Abramchuk, S.S.; Krivtsov, G.G.; Ezhov, A.A.; Podrugina, T.A.; Klyachko, N.L.; Beklemishev, M.K. Imaging-Guided Delivery of a Hydrophilic Drug to Eukaryotic Cells Based on Its Hydrophobic Ion Pairing with Poly(hexamethylene guanidine) in a Maleated Chitosan Carrier. Molecules, 2021, 26, 7426. https://doi.org/10.3390/molecules26247426
The work is aimed at the search of selective fluorescent sensors without using specific artificial receptors, antibodies, enzymes etc. With this end in view, methods based on non-covalent binding of target analytes are sought. We observed dramatic changes in the emission spectrum of a carbocyanine dye in a micellar surfactant solution (cetyltrimethylammonium bromide, CTAB) in the presence of dipyrone (metamizol, analgin): the 480 nm band intensity increases with a simultaneous decrease in intensity in near-IR region (720 nm). MALDI and NMR-1H data show the intact molecules of dipyrone and the dye. The detection can be performed in the presence of other organic species and inorganic salts. Dipyrone testing is feasible within 5×10–7...5×10–4 M with RSDs of 3.5% by using a visualizer instead of a spectrofluorimeter.
Sofia A. Zakharenkova, Anna S. Kriveleva, Ekaterina A. Katkova, Irina A Doroshenko, Nikolai Yu. Polovkov, Tatyana A. Podrugina and Mikhail K. Beklemishev. Non-covalent binding and selective fluorescent sensing of dipyrone with a carbocyanine dye and cetyltrimethylammonium bromide. Methods Appl. Fluoresc. 2021, 9, 015001. DOI: 10.1088/2050-6120/abc13c
Fluorescence fingerprinting
A variant of the “fingerprinting” method is proposed, based on a change in the fluorescence intensity of a mixture of fluorophores of different nature (“fluorescence tongue”). Water-soluble quantum dots (CdSe / CdS / ZnS, AgS, PbS, CuInS2, ZnS), and organic fluorescent dyes were used as fluorophores. It was found that fixation of the fluorophore on silica nanoparticles enhances the influence of model analytes on the fluorescence intensity (presumably due to sorption of the analyte and ensuring its proximity to the fluorophore). For use in the developed method, mixtures of up to five fluorophores were composed; in such a mixture, fluorophores can significantly quench each other's fluorescence. Medicinal substances (antibiotics of various classes, sulfonamides, etc.) were used as model analytes. Analytes have different effects on the fluorescence of different fluorophores. At the same time, emission of a mixture of fluorophores is quenched by analytes differently than individual fluorophores. For four substances (amikacin, piracetam, sulfamethoxazole, chloramphenicol), the ability to recognize both individual substances and their binary and ternary mixtures (substances were introduced in equal concentrations) was shown by processing the fluorescence spectra using the principal component method. Recognition efficiency was evaluated by calculating the Mahalanobis distances. Recognition of mixtures is most effective using mixtures of 3 or 4 fluorophores. An alternative to the proposed recognition method, based on obtaining the UV spectra of analyte mixtures and their processing by the principal component analysis, is less effective.
Nikolai N. Divyanin, Anastasiya V. Razina, Elizaveta A. Rukosueva, Andrei V. Garmash, Mikhail K. Beklemishev. Discrimination of 2-3-component mixtures of organic analytes by a “fluorescent tongue”: A pilot study. Microchem. J. 135 (2017) 48–54.
The possibility of the formation of a ternary compound (“sandwich”) of dialdehyde (malonic, glutaric or glyoxal) with ethylenediamine fluorescein thiocarbamyl (EDF) and silica nanoparticles modified non-covalently with polyethyleneimine (SiO2 / PEI), followed by fluorimetric determination, has been shown. The mixed Schiff base SiO2 / PEI – dialdehyde – EDF (sandwich) is formed in an acetic acid medium when heated in a water bath. The sandwich compound and the excess of SiO2 / PEI are separated from the unreacted fluorophore by centrifugation, the precipitate is washed, resuspended in water and the fluorescence of the solution is measured (λex = 470 nm, λem = 520 nm). The analytical cycle takes no more than half an hour. The detection limit of dialdehydes in pure water is 1 × 10–5 M, the dynamic range is 2 × 10–5–3 × 10–4 M (for malondialdehyde). The rrepeatability RSDs in this concentration range were 0.03–0.05 (n = 3). The determination of 3 × 10–4 M malondialdehyde is not hampered with comparable concentrations of sulfamethoxazole, sulfadiazine, piracetam and chloramphenicol and 1 × 10–5 M ceftriaxone, ceftazidime, analgin, isoniazid and amikacin; noticeable interferences come from proteins. The determination of glyoxal and glutaraldehyde in disinfectants was carried out.
I. A. Filenko, S. V. Golodukhina, L. O. Usol’tseva, E. M. Adamova, and M. K. Beklemishev. Covalent Binding and Fluorimetric Determination of Dialdehydes Using Aminated Silica Nanoparticles and Ethylenediamine Fluorescein. J. Analyt. Chem., 2017, vol. 72, No 9, pp. 977–985. DOI: 10.1134/S1061934817090040
The possibility of the selective determination of ceftriaxone in an aqueous solution by quenching the fluorescence of CdSe quantum dots (with CdS / ZnS shells) without separation is shown. The definition is based on our discovery of the formation of a molecular complex of ceftriaxone with polyethyleneimine. After binding of polyethylenimine to quantum dots lacking a stabilizer, a triple complex of ceftriaxone-polyethyleneimine-quantum dot is formed, in which the antibiotic is hydrolyzed by heating. The resulting mercapto groups quench the fluorescence of quantum dots. The metrological characteristics of the ceftriaxone determination method based on the described phenomena are similar when using a spectrofluorimeter and a camera in combination with a UV LED (Cmin = 1 × 10–6 M, sr = 0.1).
V. M. Karpov, D. V. Spektor, M. K. Beklemishev. Determination of ceftriaxone by the fluorescence quenching of quantum dots using binding with polyethyleneimine. Journal of Analytical Chemistry, 2016, Vol. 71(5) pp. 519–526. DOI: 10.1134/S1061934816050051
New approaches to the determination of polyelectrolytes, in particular, a disinfectant, a cationic polymer-oligomer poly(hexamethylene guanidine) (PHMG). The developed methods for the determination of PHMG in waters have wider ranges of linearity and lower detection limits than all known optical methods for the determination of PHMG.
Thus, a method for the determination of PHMG based on the formation of mixed PHMG aggregates with magnetic nanoparticles and CdSe / CdS / ZnS (QD) quantum dots and magnetic separation of the formed aggregates from excess QD is proposed. The advantages of using QD in comparison with carbon dots and fluorescein and the feasibility of using mercaptopropionate to stabilize QD fluorescence are shown. A method has been developed for the semi-quantitative fluorimetric determination of PHMG in waters in the form of mixed aggregates with a detection limit of 0.025 mg / L, a linearity range of 0.05 - 0.2 mg / L, and RSDs of up to 0.27. The possibility of determining the PHMG by a similar method using a LED as a UV light source and registering fluorescence with a digital camera with a light filter is shown; the characteristics of the method are close to those for a spectrofluorimeter (linearity range 0.05 - 0.2 mg / L, LOD = 0.04 mg / L and RSDs up to 0.23). Selectivity factors were obtained with respect to Na+, Ca2+, Mg2+, Cu2+, Fe3+, cationic, anionic and nonionic surfactants, humic acids, cationic and anionic polymers. The expediency of masking metal cations using EDTA is shown. The possibility of determining PHMG in wastewater, tap water and pool water is shown.
Konstantin V. Likhachev, Elena O. Ovcharenko, Alexander I. Dityuk, Sergei S. Abramchuk, Konstantin M. Efimov, and Mikhail K. Beklemishev. Fluorescent Determination of Poly(hexamethylene Guanidine) via the Aggregates it Forms with Quantum Dots and Magnetic Nanoparticles. Microchim. Acta. 2016. 183(3). P. 1079-1087.
Another method of highly sensitive determination of polyhexamethylene guanidine (PHMG) in solution is based on the aggregation of silver nanoparticles, observed by Rayleigh scattering spectroscopy. The interfering effect of surfactants, humic acids and metal cations (in the presence of EDTA) is relatively small; 0.05 M NaCl does not interfere. Other cationic polyelectrolytes - polyethyleneimine and polydiallyldimethylammonium chloride - aggregate AgNPs several times weaker than PHMG. Methods have been developed for the determination of PHMG in waters: waste water from storm sewers in Moscow in the range 4 · 10–8 ... 1.3 · 10 -5 M (calculated by monomer) with a detection limit of 2 · 10–8 M (0.004 mg / L), which is lower than the minimal allowed concentration for PHMG (0.01 mg /L); tap water in the range of 5 · 10–7 - 1.3 · 10–5 M with an LOD of 4 · 10–7 M and swimming pool water in the range of 1 · 10–5 - 6 · 10–5 M with an LOD of 6 · 10– 6 M. The methods developed on the basis of this approach are characterized by wider ranges of linearity and lower detection limits than all known optical methods for determining the PHMG.
Artemyeva A.A., Samarina T.O., Beklemishev M.K., Efimov K.M., Dityuk A.I. A method for quantitatively determining the concentration of polyhexamethylene guanidine hydrochloride in an aqueous solution. Russian patent No. 2557930. The priority of the invention of 06/04/2014. Registered in the State register of inventions of the Russian Federation 06/30/2015.
Artemyeva A.A., Samarina T.O., Sharov A.V., Abramchuk S.S., Ovcharenko E.O., Dityuk A.I., Efimov K.M., Beklemishev M.K. Highly Sensitive Determination of Poly(hexamethylene Guanidine) by Rayleigh Scattering Using Aggregation of Silver Nanoparticles. Microchim. Acta, 2015, vol. 182 (5–6), pp. 965–973. doi: 10.1007/s00604-014-1411-6.
The possibility of using the Rayleigh scattering method to determine quercetin in an aqueous solution using silver nanoparticles (NPs) is shown. It was found that low concentrations of quercetin (of the order of μg / L) significantly increase the intensity of Rayleigh scattering of a solution of NSF stabilized by cetyltrimethylammonium bromide (CTAB) or sodium n-dodecyl sulfate (DDS). The increase in scattering is explained both by the growth of nanoparticles (an increase is observed only in the presence of an excess of AgNO3) and by the aggregation of NPPs with quercetin (which is confirmed by the results obtained by dynamic light scattering). The conditions for determining quercetin in an aqueous solution were selected; detection limits are 0.01 and 0.03 μM, linear ranges are 0.1-1.3 and 0.1-2.0 μM for DDS and CTAB-stabilized NPs, respectively; relative standard deviation does not exceed 7%. Interestingly, some other bioflavonoids (rutin, dihydroquercetin and naringenin) do not alter the quercetin signal and do not interfere with its determination in the equimolar ratio (0.5 μM each). Other compounds (asparagine, uric acid, urea and some inorganic ions) do not interfere with the determination in large quantities.
Liliya O. Usoltseva, Tatiana O. Samarina, Sergei S. Abramchuk, Aleksandra F. Prokhorova, Mikhail K. Beklemishev. Selective Rayleigh Light Scattering Determination of Trace Quercetin with Silver Nanoparticles. Journal of Luminescence, 2016, vol. 179, pp. 438-444.
A method for determining the antibiotics of the aminoglycoside series based on the covalent binding of an analyte to a water-soluble polymer with subsequent measurement of the intensity of Rayleigh light scattering (λex = λem) is proposed. The determination was carried out using the example of amikacin, the amide bond of which with the copolymer of 4-styrenesulfonic and maleic acids was formed by the carbodiimide method. The conditions for carrying out this reaction were selected to obtain the maximum scattering intensity of the product (time 15 min, pH 7.0, ionic strength 0.02, measurement of scattering at 362 nm). The determination of amikacin is not affected (multiple ratios, mol / mol) by: Al(III), Co(II), Cu(II), Fe(III), Ni(II), Zn(II) - 300, Cl– and NO3 - 250, Ca(II) and Mg(II) 50, HCO3– 20; nitrogen-containing compounds affect at 20-50-fold ratios, protein - 2.5-fold. The range of detectable concentrations is 0.2–35 mg/L, LOD = 0.08 mg/L.
Samarina T.O., Beklemishev M.K. Determination of amikacin by Rayleigh scattering after covalent binding of the analyte to a water-soluble polymer. Moscow University Bulletin. Ser. 2. Chemistry. 2015. Vol. 56. No. 5. P. 25–31.
Molecular Imprinting
A method is proposed for producing molecular imprints of polar organic templates in the form of complexes with a polar compound in a non-polar polymer matrix. Such a scheme should minimize the formation of sites of non-selective binding of the template. As a model template, bromothymol blue was used as a complex with polyethyleneimine, which was introduced into the photopolymerizable mixture of styrene - divinylbenzene on a track-etched membrane as a substrate. The method of diffusion through imprinted membranes shows a high selectivity of the obtained imprints with respect to substances structurally dissimilar to the template (4-nitrophenol and neutral red) and low to other sulfophthalein dyes. The approach is promising for obtaining selective molecularly imprinted membranes and sorbents.
A variant of molecular imprinting based on carbodiimide crosslinking of amino acids in the presence of a cationic polyelectrolyte (polyethyleneimine) is proposed. A dye (bromothymol blue) was used as a template, which facilitated the control of the presence of a template in the polymer phase, and track-etched membranes preoxidized and activated by carbodiimide / pentafluorophenol were used as a substrate. Membranes imprinted using only one amino acid (leucine) bind the template more strongly than using a mixture of amino acids. The template diffuses through the imprinted membranes faster than through the unprinted ones, and the imprinted silica absorbs the template more completely than the unprinted one (imprinting factor is 2–4).
When determining phytosteroids in plant materials by HPLC-MS, pre-concentration of analytes is useful, for which it would be convenient to use molecularly imprinted sorbents. However, phytosteroid molecules do not have ionizable groups; therefore, obtaining their molecular imprints is difficult. We have proposed methods for molecular imprinting of phytosteroids (using ecdysten, or 20-hydroxyecdysone, as example) into titanium dioxide, based on the interaction of ecdysten with titanium n-butoxide, followed by fixing the obtained precomplex on the surface of a TiO2 nanopowder and washing out the template. Samples of imprinted sorbents retain a greater amount of ecdysten compared to similar sorbents without molecular imprints (imprinting factor is about 2).
Sevko D.A., Abramchuk S.S., Ikhalainen A.A., Antokhin A.M., Taranchenko V.F., Goncharov V.M., Aksenov A.V., Mitrofanov D.A., Sinitsyn M Yu., Beklemishev M.K. Selective extraction of phytoecdysteroids from a plant extract with a molecularly imprinted sorbent based on titanium dioxide // Chemistry of plant raw materials. 2015. No 2. P. 59–68. DOI: 10.14258/cprm.201502542. http://journal.asu.ru/index.php/cw/article/view/542/588
The regularities of the formation of the analytical signal in quantum dot - polyelectrolyte - analyte - carrier systems are studied. We studied the fluorescence of quantum dots (QDs) of CdTe and CdSe with various shells stabilized in an aqueous solution by dimercaptosuccinate and acrylic acid copolymers. It was found that cationic polymers (polyallylamine, polydimethyldiallylammonium chloride) at concentrations of ~ 0.001 M reduce the fluorescence intensity of QDs by 10–20%, and at concentrations of ~ 0.01 M they increase it. In addition, cationic polymers restore the quantum yield of QD luminescence, the emission of which is partially quenched by a buffer solution (Tris-buffer and acetate). Antibiotics of the fluoroquinolone and cephalosporin series, tetracycline, and some other low molecular weight nitrogen and sulfur compounds (a total of 17 compounds) were studied as model analytes. It was found that some of the compounds quench the fluorescence of QDs at a concentration of no higher than 1 · 10–4 M. The most powerful quenchers are tetracycline, isoniazid, penicillin V, and fluoroquinolones. Individual compounds increase the fluorescence quantum yield (sulfamethoxazole). In the presence of cationic polyelectrolytes (polyethyleneimine, polyallylamine), part of the compounds diminishes the quenching effect, which can be explained by sorption of the polycation on the quantum dots and the difficulty of access of the quencher to the core. The QDs were deposited on silica (plates 'Sorbfil' for TLC) by sorption from a solution and the effect of the same model analytes on quenching of QDs on the carrier was studied. It was found that the range of compounds extinguishing fluorescence becomes narrower. In addition, in some cases, the modification of QDs with a cationic polymer leads to an increase in the quantum yield of fluorescence; so, tetracycline increases it 5-fold (at a concentration of 1 × 10–4 M).
A method for determining polyelectrolytes based on their compensation of the charge of a polyelectrolyte multilayer deposited on a screen-printed electrode is proposed. The charge state is controlled by the current of hexacyanoferrate(III) diffusing through the multilayer to the electrode. Externally, the influence of the analyte looks like the restoration of the shape of the current-voltage curve, changed due to preliminary sorption of the opposite charge on the polyelectrolyte-modified electrode. Before determination, the screen-printed electrode is treated with polyelectrolyte, after which the cyclic voltammograms are recorded in the range from –1000 ... +1000 mV at a speed of 100 mV / s. The difference potential of the peak of reduction (or oxidation) of hexacyanoferrate in the analyzed solution and in the background solution is used as an analytical signal (the voltammogram of this background solution should be recorded after the electrode is immersed in the anionic polyelectrolyte solution, if a cationic one is determined). The detection limits for cationic polyelectrolytes by this method are n · 10–5 M (calculated as a monomer), RSD = 8% at 1 × 10–4 M (n = 3). The determination is not interfered with ~ 0.1 M NaCl, as well as calcium and magnesium salts in the concentrations present in natural waters. Surfactants that also restore the shape of current-voltage curves (at a concentration of 1 · 10–5 M) interfere with the determination. The disadvantage of this methodology is the incompleteness of electrode regeneration after measurement and the limited number of regeneration cycles.
The possibility of molecular imprinting of low molecular weight organic compounds into polyelectrolyte multilayers is shown on the example of penicillin V and 20-ecdysterone as templates, polyallylamine and polyacrylic acid as polymers and epichlorohydrin and glutaraldehyde as crosslinkers. The substrate for the deposition of multilayers was a polyethylene terephthalate track-etched membrane, the permeability of which to templates after modification was studied by passive diffusion. It was shown that the deposition of multilayers on a substrate is possible not only from water, but also from an organic solvent (e.g., ethanol). The introduction of an ionic liquid into the solutions from which polyelectrolytes precipitate leads to an increase in the permeability of the resulting membrane, which is probably caused by a change in the conformation of the polymer in the presence of ionic liquid.
Lokhova L.O., Polyanina D.A., Beklemishev M.K. Molecular imprinting of penicillin V in layers of polyelectrolytes deposited on a track membrane. // Vestn. Mosk. un-that. Ser. 2. Chemistry. 2014.V. 55. No. 1. P. 24–28.
Membranes with molecular imprints were obtained, that are not yet sufficiently used in separation methods. The method of molecular imprinting is interesting by thatit allows one to obtain membranes with increased permeability to the template and selective for it. Bioflavonoids (rutin and naringenin) were used as templates, PETP track-etched membranes (pore diameter 0.4 μm) were used as a substrate for polymerization, methacrylamide was the monomer, triethylene glycol dimethacrylate was the crosslinker, and dimethylformamide was the porogenic solvent. The polymerization was carried out under irradiation with UV light (the initiator was Darokur 1173 by Ciba) in various ways (with and without air access restriction). The polymerization was carried out for 1 h, washing the template requires 30 min. Of the functional monomers, acrylic acid is the most effective; to improve the quality of the imprint, it is advisable to introduce transition metal salts (copper(2+) or nickel(2+)) into the polymerization mixture. After polymerization, the template was washed from the membrane with acetone. The diffusion transfer of the template and other compounds through the resulting membranes was studied. We chose the polymerization conditions under which the diffusion rate across the imprinted membrane of the template was maximum and of other compounds minimum. It is possible to carry out selective diffusion of bioflavonoids not only from organic, but also from aqueous and aqueous-organic media. A rutin imprinted membrane shows a selectivity factor of SF = 17 against naringenin (SF is the ratio of rutin and naringenin transfer rates), while a naringenin imprinted membrane allows it to be completely separated from quercetin (SF = 9). analyte diffusion rates are close to the maximum possible. The maximum separation factors for bioflavonoid pairs in the separate transfer mode are of the order of 5–15, which is comparable with the results achieved by foreign researchers. The transfer of ascorbic acid through the resulting membranes was also studied. Thus, by varying the polymerization conditions, it is possible to obtain imprinted membranes for the selective isolation of bioflavonoids. The advantages of imprinting in membranes compared to bulk polymerization are shorter polymerization time (no more than 1 hour), faster template washing (several minutes); the advantage of using membranes as a means of separation compared to using a powdered sorben: it is not necessary to crush and fractionate the resulting polymer, with membrane separation, it is not necessary to desorb the analyte from the sorbent. Membrane diffusion is used to isolate bioflavonoids from plant samples (onions, dill, immortelle).
Galina A. Kryvshenko, Pavel Yu. Apel, Sergei S. Abramchuk, and Mikhail K. Beklemishev. A highly permeable membrane for separation of quercetin obtained by nickel(II) ion-mediated molecular imprinting. Sep. Sci. Technol. 2012. Vol. 47. No 12. P. 1715–1724.
http://www.tandfonline.com/doi/abs/10.1080/01496395.2012.659317
G.A. Kryvshenko, M.K. Beklemishev. The method of separation of quercetin from an aqueous solution. Application for a patent of the Russian Federation No. 2011119249 of May 16, 2011. Patent of the Russian Federation No. 2458921 C1.
For imprinted membranes obtained by polymerization of acrylic monomers with a crosslinker, higher permeability was found in the case of imprinting not of an individual organic template, but of its complex with another compound. It has been suggested that imprinting of the complex produces molecular imprints that bind the template only in part of the active groups. This leads to a less durable and more reversible binding, which allows one to observe the diffusion of the template through the membrane (studied by the example of penicillin V).
A method is proposed for determining the antioxidant activity (AOA) of individual compounds and plant extracts using oxidation reactions of 3,3 ', 5,5'-tetramethylbenzidine (TMB) with periodate and persulfate. Two versions of the methodology have been developed: one based on preliminary preparation of a colored TMB oxidation product and subsequent measurement of AOA analyte to reduce the absorption of this product (the "equilibrium option") and one based on measuring the formation rate of the TMB oxidation product, which slows down in the presence of reducing agents (the "kinetic version") . The data obtained by the proposed methods and using the standard method of decolorization of the ABTS radical cation are close, which confirms the efficiency of the proposed methods. Substances that do not have antioxidant activity, as well as proteins, do not interfere with the determination of AOA. The sensitivity of the technique is not inferior to the well-known ones, including those based on the reduction of the ABTS radical cation. The technique is simple and rapid (it takes 5 minutes to determine, no more than 20 minutes to prepare the reagents, unlike ABTS based methods, where it takes at least 6 hours). When determining AOA of samples of animal origin, one can use both the kinetic and equilibrium versions of the method; for plant extracts, it is desirable to use the equilibrium option. A test version of the method is proposed with visual observation of the color on filter paper, with which it is possible to measure AOA directly in the Trolox equivalent and allows the use of small volumes of diluted plant extracts (100 μl).
G.A. Kryvshenko, M.K. Beklemishev. determination of antioxidants using oxidation reactions of 3.3 ', 5.5'-tetramethylbenzidine with persulfate and periodate // Chemistry of plant raw materials. 2011. No 1. S. 123–128.
Polyethylene terephthalate track-etched membranes were studied as a separation means. A number of features of the transport of organic compounds through an unmodified membrane (thickness 10 μm, pore diameter 0.03 μm) were discovered: the difference between the state of compounds in pores and the state in the bulk of the solution; concentration of the compound in the pores of the membrane (mainly in the solution phase); transfer of an organic compound by the diffusion potential of inorganic salts. The presence of non-ion-exchange selectivity (e.g., in phenol / 2-naphthol, benzene / benzyl alcohol vapors) of unmodified membranes was revealed. Track-etched membranes modified by polyelectrolyte complexes of polyethyleneimine (PEI) and polyanethol sulfonic acid (PAS) were obtained, and the features of their permeability were found: a sharp dependence of permeability on the number of layers of polyelectrolytes; anisotropy (with asymmetric modification); an increase in the transfer selectivity factor of the most rapidly transferred compounds (10–100 times in comparison with an unmodified membrane). The possibility of selective isolation of aniline in the presence of model compounds is shown.
A. Belkova, A. I. Sergeeva, P. Y. Apel, M. K. Beklemishev. Diffusion of aniline through a polyethylene terephthalate track-etched membrane // Journal of Membrane Science. V. 330. No 1-2. Pp. 145-155.
Work continued on the study of track-etched membranes modified with polyelectrolyte multilayers (PEMs) as a means of separation and concentration of organic compounds. The structural features of such modified membranes were revealed: with a sufficient number of polyelectrolyte layers, the pores are completely filled with the gel of the polyelectrolyte complex, which has a loose structure, not only preserving permeability with respect to diffusing compounds, but also more permeable for individual substances compared to the initial substrate. In membranes modified with layers of polyethyleneimine and polyanethol sulfonic acid, the transfer of aromatic amines is accelerated, the selectivity of which is not explained solely by the size or charge of the particles. The acceleration of transfer can be associated with the formation in the gel structure of the polymer multilayer domains, oriented along the pores. The role of the nature of cationic and anionic polyelectrolytes was studied (polyanetholsulfonic acid is more effective than polystyrenesulfonic acid; cationic polymers can be different). In addition, TEMs based on complexing polymers (polyethyleneimine) can trap transition metal ions without passing them through the membrane, but allowing diffusion of organic compounds.
Kinetic methods of analysis
To expand the range of compounds determined by kinetic methods, it was proposed to carry out a targeted choice of indicator reaction - analyte systems based on an analysis of the literature data on the interaction of radical particles with organic compounds. Based on the information that the hydroxyl radical interacts with polyols, it was suggested that sugars can influence the reaction rate at which the hydroxyl radical is generated. The oxidation of 3.3 ', 5.5'-tetramethylbenzidine (TMB) Н2О2 in the presence of iron (+2, +3) was used as such a reaction. To increase the selectivity, the determination was combined with preliminary separation by TLC. When carrying out the reaction on silica wafers, among the 19 model compounds studied, the greatest inhibitory effect is exerted by benzoic acid and glucose, typical hydroxyl radical scavengers. A technique has been developed for the semiquantitative determination of glucose in a thin-layer chromatogram. Glucose is detected as a lighter spot on a blue-green background (Rf ~ 0.5). The calibration graph was built as a reflection coefficient, measured on an OTDR, depending on the logarithm of the analyte concentration. Semi-quantitative determination of glucose is possible in the range of 1 · 10–5–0.01 M. The determination of glucose in drinks and saliva is carried out.
The second example of application of this approach was our proposed bromination reaction of methyl violet N-bromosuccinimide (NBS). Among model analytes, benzyl alcohol, aniline, benzylamine, cysteine, methylamine, 1-naphthylamine, thiourea slow down this reaction, while sodium benzoate, acrylamide, p-benzoquinone, triethylbenzylammonium chloride do not affect the reaction. Of particular interest is the ability to determine benzyl derivatives for which kinetic methods are not described. It was shown that ~ 10–6 M of benzyl alcohol and 10–4 M of benzylpenicillin can be determined by this indicator reaction.
As part of a series of works on expanding the range of compounds determined by kinetic methods and the targeted selection of indicator-analyte systems, it was proposed to use the rhodamine 6G bromination reaction as an indicator for determining brominating organic compounds. Based on a literature analysis, organic compounds capable of easily brominating were selected (benzyl alcohol, benzylamine, benzoquinone, hydroquinone, acrylamide, benzylpenicillin). It was found that during the interaction of rhodamine 6G with N-bromosuccinimide, the band of the rhodamine fluorescence spectrum (590 nm) shifts to the short-wavelength region (537 nm), which can be explained by the formation of rhodamine bromine derivatives and is convenient for measuring the reaction rate.
The oxidation of 1,2,4-triacetoxybenzene (pyrogallol “A”) and 3,3 ', 5,5'-tetramethylbenzidine (TMB) with air oxygen was studied under irradiation with visible light in the presence of tris (2,2'-bipyridylate) ruthenium ( II) (Ru (bipy) 32+) as a photosensitizer. Pyrogallol “A” photooxidation is selectively inhibited by the benzoate ion at pH 9–11. Other carboxylic acids do not affect the reaction rate; short-chain aliphatic amines and transition metal ions affect it. A method for the semi-quantitative determination of benzoate on a paper chromatogram has been developed. As a reagent for the manifestation, a solution of pyrogallol A and a photosensitizer are used; after spraying with which the chromatogram is irradiated with an incandescent lamp. Benzoate is detected as a bright spot with Rf ~ 0.3 on a brown background. Measurement of the reflection intensity of the spot on an OTDR allows one to determine 1 · 10–5–0.01 M benzoate with an accuracy of half-order. Benzoate in drinks was determined at a level of 10–4–10–3 M; capillary electrophoresis was used as a comparison method.
L.Yu. Belyaeva, A.F. Prokhorova, M.K. Beklemishev. Determination of the benzoate ion by paper chromatography with detection by the inhibitory effect in the reaction of photosensitized autooxidation of pyrogallol "A". J. Anal. Chem., 2010, 65(1), p. 66–72
The oxidation of 2,2'-azino-bis (3-ethyl-benzthiazolin-6-sulfonic acid) (ABTS) periodate was studied, including in the photochemical version in the presence of Ru (bipy) 32+ as a photosensitizer. The inhibitory effect of Cd(II), Ni(II), L-cysteine, quercetin and the accelerating effect of Mn(II), Ni(DMG)2 (DMG - dimethylglyoximate) and diethylamine on the reaction rate ABTS - periodate were found. The dark ABTS - periodate reaction was shown to be similar to the TMB - periodate reaction in a set of influential compounds. The metals that influence the TMB – IO4– reaction (Cd(II), Ni(II), and Mn(II)) do not affect the photoreaction rate of ABTS – IO4–. Thus, the photochemical variant of the indicator reaction is selective in relation to organic compounds; quercetin (10–7 M) and L-cysteine (10–6 M) have the greatest inhibitory effect.
Past years results
The foundations of a new sorption-kinetic analysis method based on conducting indicator reactions on the surface of carriers are proposed. It was suggested to use a combination of the sorption preconcentration of analytes with their determination directly on the solid carrier as a general approach to increasing the selectivity and sensitivity of kinetic methods of analysis. A number of new indicator reactions were proposed for the determination of organic and analytes and metal ions. The feasibility of determining the analytes by the kinetic method in the permeate and membrane surface after preconcentration using track-etched membranes was shown. Kinetic methods for the determination of synthetic water-soluble polymers were developed.
2013-2020 © Laboratory of Kinetic Methods of Analysis, Department of Chemistry, M.V. Lomonosov Moscow State University  To the top
To the top



